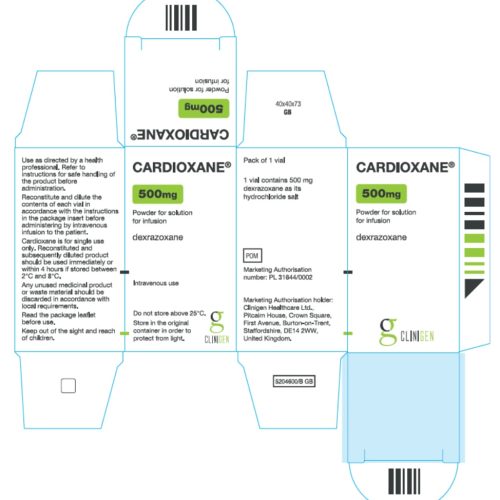
Cardioxane 500 mg Powder for Solution for Infusion
Cardioxane 500 mg is indicated for reducing the incidence and severity of cardiomyopathy associated with anthracycline chemotherapy in adult patients. FDA approved brand name Cardioxane is not (yet) registered or available in India. The access to the drug is provided as per Name Patient Program respecting the regulatory framework as desired by local authorities.
Home | Cardioxane Injection
Submit a request to access drug used to treat, diagnose or prevent serious or life-threatening conditions.We’ll get back to you within 1 business days!
For Indian Patient Enquiry under NPP.
For any urgent inquiries or assistance needed, please reach out to our Support Team. Rest assured, we will respond within 24 hours, from Monday to Saturday, during the hours of 9:00 A.M to 18:00 PM.
Cardioxane 500 mg Powder for Solution
Dexrazoxane is a cardioprotective agent used to reduce heart damage (cardiomyopathy) caused by anthracycline chemotherapy (e.g., doxorubicin) in breast cancer patients.
Cardioxane 500 mg Powder for Solution for Infusion contains the same active substance, dexrazoxane hydrochloride, as FDA-approved Zinecard. It is available as 500 mg/vial (no 250 mg/vial presentation is available). It is a clinically acceptable substitute for the out of stock U.S. Dexrazoxane for Injection products. Cardioxane is available only by prescription in the U.S.
In the U.S., the labelling for Dexrazoxane Injection (Zinecard) includes the following indication: ZINECARD is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy.
The United States Food and Drug Administration has also approved a dexrazoxane for use as a treatment of extravasation resulting from IV anthracycline chemotherapy. Extravasation is an adverse event in which chemotherapies containing anthracylines leak out of the blood vessel and necrotize the surrounding tissue.
Drug (Brand / Generic): Cardioxane® 500 mg Powder for Solution for Infusion
Current Indication: cardiomyopathy
Approval Date: Cardioxane (dexrazoxane) 500 mg powder for solution for infusion has key regulatory dates in the EU and US. An EU-wide decision on its updated, restricted license was implemented on July 19, 2017. In the U.S., it was listed for import via FDA-authorized shortage distributors on July 28, 2017
Cardioxane® 500 mg Powder for Solution for Infusion
Dexrazoxane (as its hydrochloride salt) Intravenous use for short infusion.
Do not store above 25°C. Store in the original container. Protect from direct sunlight.
For more highlights of prescribing information regarding Indications and usage, dosage and administration, dosage forms and strengths, patient counseling information and medication guide. Click Here
Alleviare Life Sciences
About Us
Alleviare Life Sciences Pvt. Ltd. (India) serves as a facilitator for medical and pharmaceutical products, commencing its operations in 2005. Headquartered in Delhi, India, Alleviare operates nationwide, providing a range of comprehensive services to patients, doctors, hospitals, and government healthcare facilities.
Our specialization lies in facilitating access to medicines, leveraging our expertise in sourcing and providing documentation assistance for programs such as Named Patient Program, Managed Access Program, and Early Access Program. We take pride in ensuring smooth and efficient access to medications for those who require them.

Alleviare India is a trusted pharmaceutical access facilitator in India, aligned with Good Distribution Practice (GDP) standards. We provide specialized support to help patients and healthcare professionals access Cardioxane 500 mg, an internationally approved therapy.
Where VYVGART is not commercially available in India, we facilitate access through regulated pathways such as the Named Patient Supply (NPS) framework. Each request is carefully managed in compliance with applicable regulations and requires a valid prescription and, where necessary, import authorization.
Get Access to FDA approved Cardioxane 500 mg in India.
We can help patients access approved Cardioxane 500 mg in the following cities across India – Ahmedabad, Bangalore, Surat, Kolkata, Pune, Indore, Nagpur, Agra, Kanpur, Patna, Coimbatore, Kochi, Delhi, Gurugram, Amaravati, Itanagar, Dispur, Patna, Raipur, Panaji, Gandhinagar, Chandigarh, Shimla, Ranchi, Bengaluru, Thiruvananthapuram, Bhopal, Mumbai, Imphal, Shillong, Aizawl, Kohima, Bhubaneswar, Jaipur, Gangtok, Chennai, Hyderabad, Agartala, Lucknow, Dehradun, Gairsain. Aizawal, Aurangabad, Barshi, Cachar, Dibrugarh, Karungapally, Kollam, Meghalaya, Mizoram, Sikkim, Pondichery, Punjab, Tripura, New Delhi, Agra, Gurugram, Alwar, Mathura, Panipat, Faridabad, Noida, Sonipat, Rohtak, Karnal, Ambala, Ghaziabad.Cardioxane 500 mg – Sourcing & Access Support
Alleviare India provides assistance in facilitating access to Cardioxane 500 mg, a prescribed oncology medication, through trusted international supply channels. We support patients and healthcare providers in identifying appropriate supply options for medicines that may have limited local availability, in accordance with applicable regulations.Access is provided only against a valid prescription, and services are offered under the Named Patient Supply (NPS) framework. Alleviare India may also assist in connecting with authorized supply channels where appropriate.Pricing & Availability in India
For information on the availability and indicative pricing of Cardioxane 500 mg, please contact us directly.The cost of the medicine is determined by the manufacturer. Additional charges may include Named Patient support service fees, shipping costs, and applicable local taxes, in accordance with regulatory requirements.Is Cardioxane 500 mg available in India?
Meet Our Medicine Access Expert for availability of Truqap medications.At Alleviare, we prioritize transparency while supporting patients, healthcare professionals, and institutions in accessing genuine medicines through trusted global sources in Canada, Europe, the USA, and Australia. We provide tailored assistance to facilitate access to prescribed therapies, including those not readily available locally, in full compliance with applicable regulatory requirements.Access is facilitated under the Named Patient Supply (NPS) framework. We also offer guidance on the availability and indicative pricing of prescribed medications across major cities in India, including Mumbai, Kolkata, Hyderabad, Chennai, Ahmedabad, Delhi, Bangalore, and Pune.Medicine: Cardioxane 500 mg (subject to prescription and regulatory guidelines)Start Your Access JourneyEmail: info@alleviareindia.com, Mobile: +91 9818274099 / 9289711088Is Cardioxane 500 mg available in India?Yes, FDA approved Cardioxane 500 mg is not (yet) registered or available in India but on request Indian patient can buy Cardioxane 500 mg at the lowest price.What documents are required to import Cardioxane 500 mg to India?
To import Cardioxane 500 mg, patients or government hospitals can place an order on behalf of the patients. The following documentation is required for the import process:- Valid prescription from a qualified doctor.
- Patients’ diagnostic reports.
- Patients’ ID proof issued by the Government of India.
- Valid prescription from the doctor.
- Import permit, if applicable.
